a PG antagonist as well. Caffeine and metoclopramide are used in combination with
analgesics and ergotamine in the treatment of migraine attacks, but controlled clinical studies on fixed combinations with analgesics are rare. The effects of orally given tolfenamic acid(200 mg), caffeine (100 mg), metoclopramide (10 mg), tolfenamic acid + caffeine (200 mg +100 mg), tolfenamic acid + metoclopramide (200 mg + 10 mg) and placebo were studied in
49 migraine patients (3 men, 46 women) in a double-blind randomized cross-over study comprising 482 migraine attacks. The patients were allowed to take either one or two capsules of each preparation for an attack. Additional drugs were allowed after 3 h. Parameters characterizing the effects and side-effects of the drugs were registered. Tolfenamic acid and its combinations were found to be effective in the treatment of acute migraine, but caffeine and metoclopramide alone did not differ from placebo. Combination with metoclopramide was better than tolfenamic acid alone as judged by the smaller dose needed and the intensity of attack. Between tolfenamic acid alone and its caffeine combination there were no statistically significant differences. o A nalgesic combinations,
PMID: 6984358
㈵ b
Tolfenamic acidとカフェインの片頭痛における治療
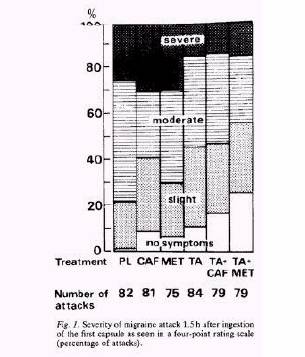
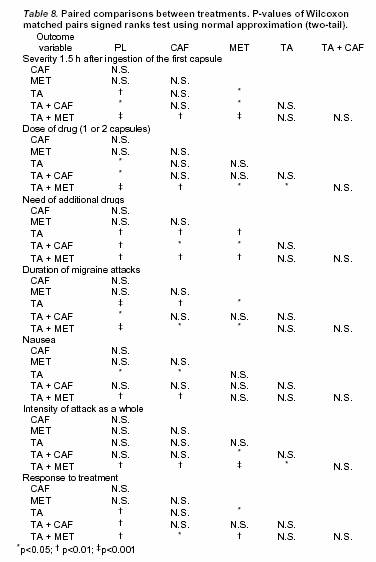
Tolfenamic acidとの併用療法では片頭痛急性期治療として有効性は高かったが,カフェイン,メトクロプラミドの単独療法ではその効果にプラセボとの差は認められなかった.
Tolfenamic acid単独とカフェイン併用では統計学的には差はなかった.
副作用は 482名中で軽度のものが47名に認められた.副作用の出現には各治療薬間での差はなかった.副作用としての嘔気出現はほんの7%にすぎなかった.
Publication Types:
• Clinical Trial
• Randomized Controlled Trial
MeSH Terms:
• Adult
• Anthranilic Acids/administration & dosage*
• Anti-Inflammatory Agents, Non-Steroidal/administration & dosage*
• Caffeine/administration & dosage*
• Comparative Study
• Drug Combinations
• Female
• Human
• Metoclopramide/administration & dosage
• Middle Aged
• Migraine/drug therapy*
• Prostaglandin Antagonists/administration & dosage*
• Pyridoxine/administration & dosage
• Random Allocation
Substances:
• Anthranilic Acids
• Anti-Inflammatory Agents, Non-Steroidal
• Drug Combinations
• Prostaglandin Antagonists
• tolfenamic acid
• Metoclopramide
• Caffeine
Pyridoxine